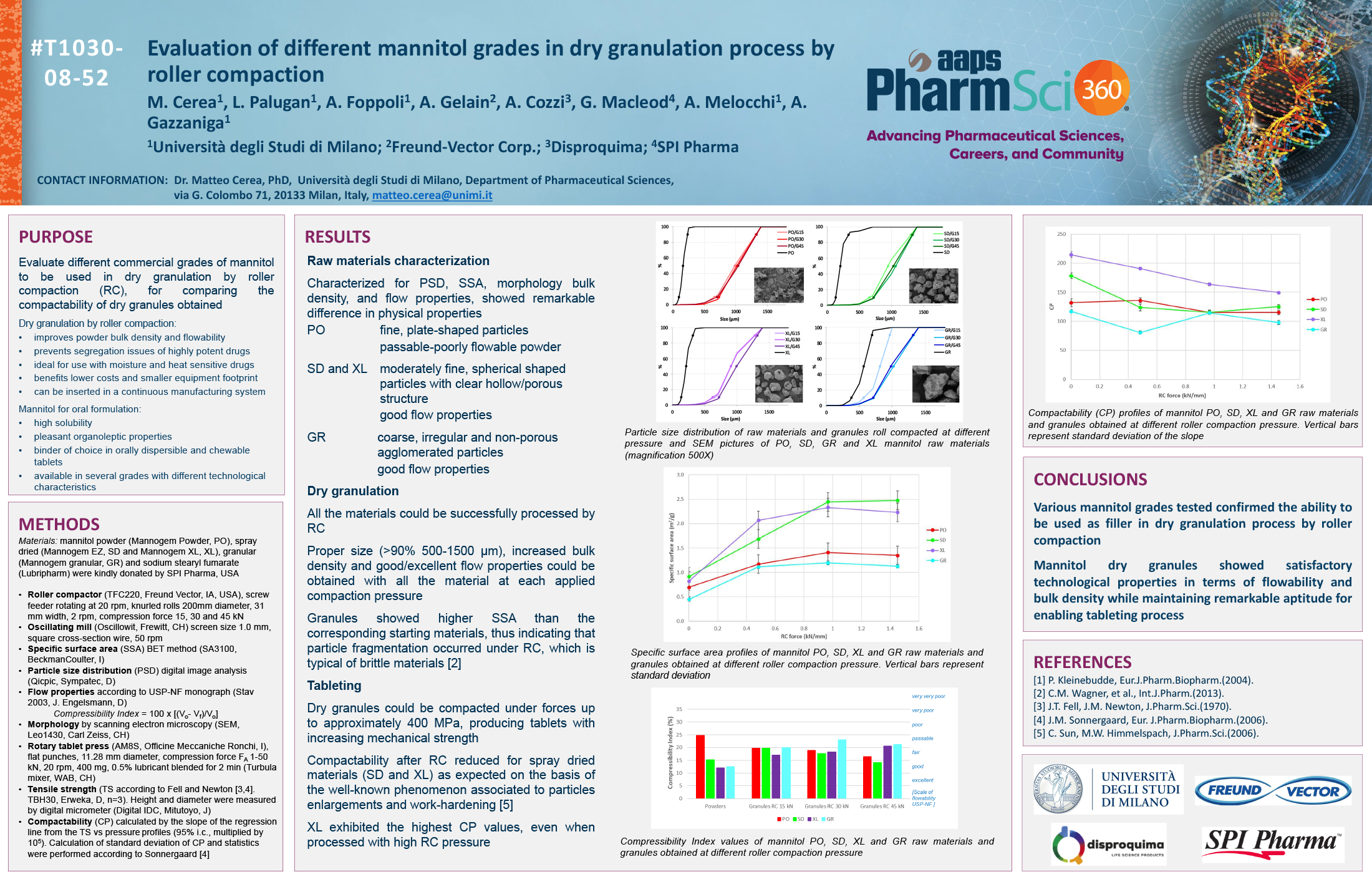
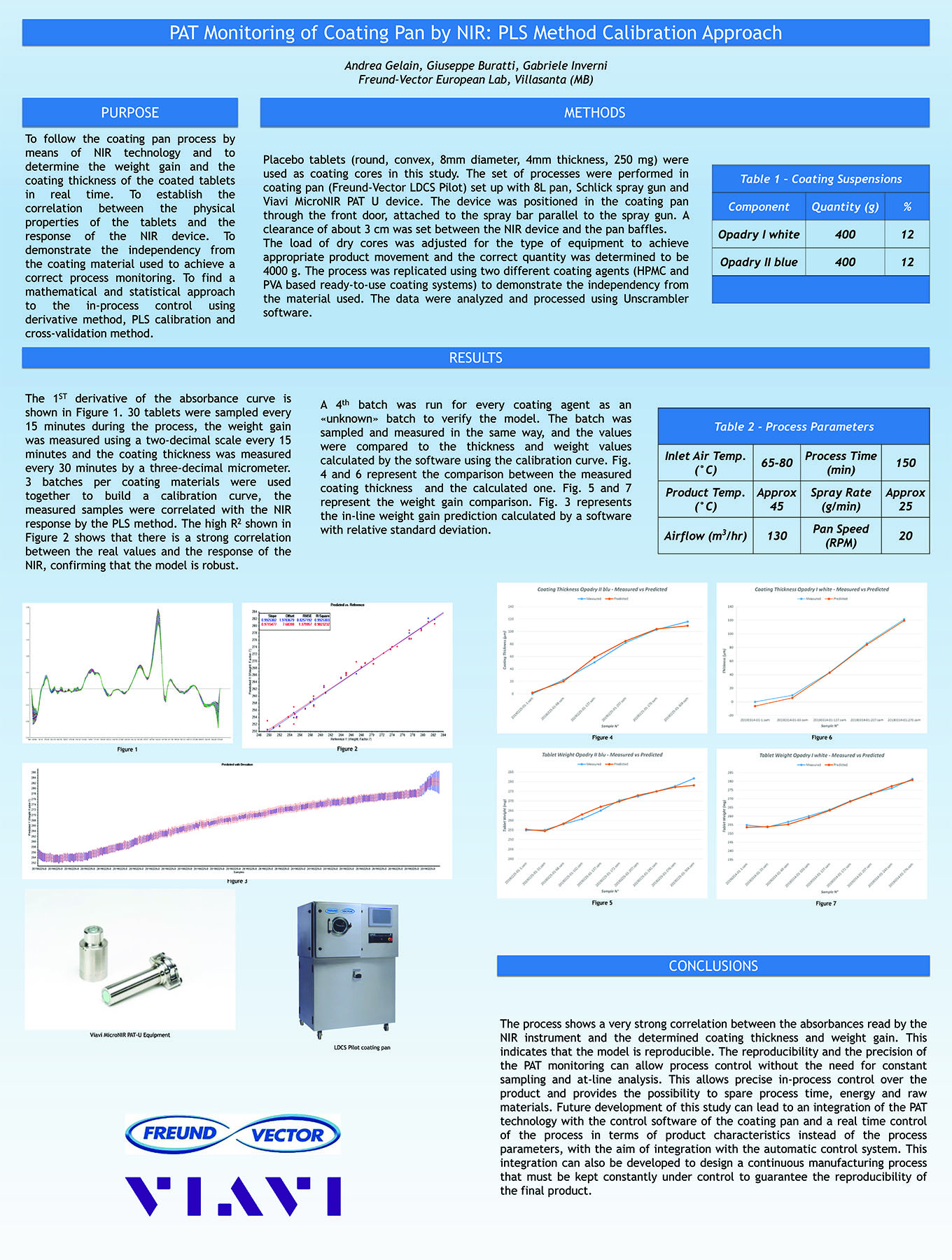
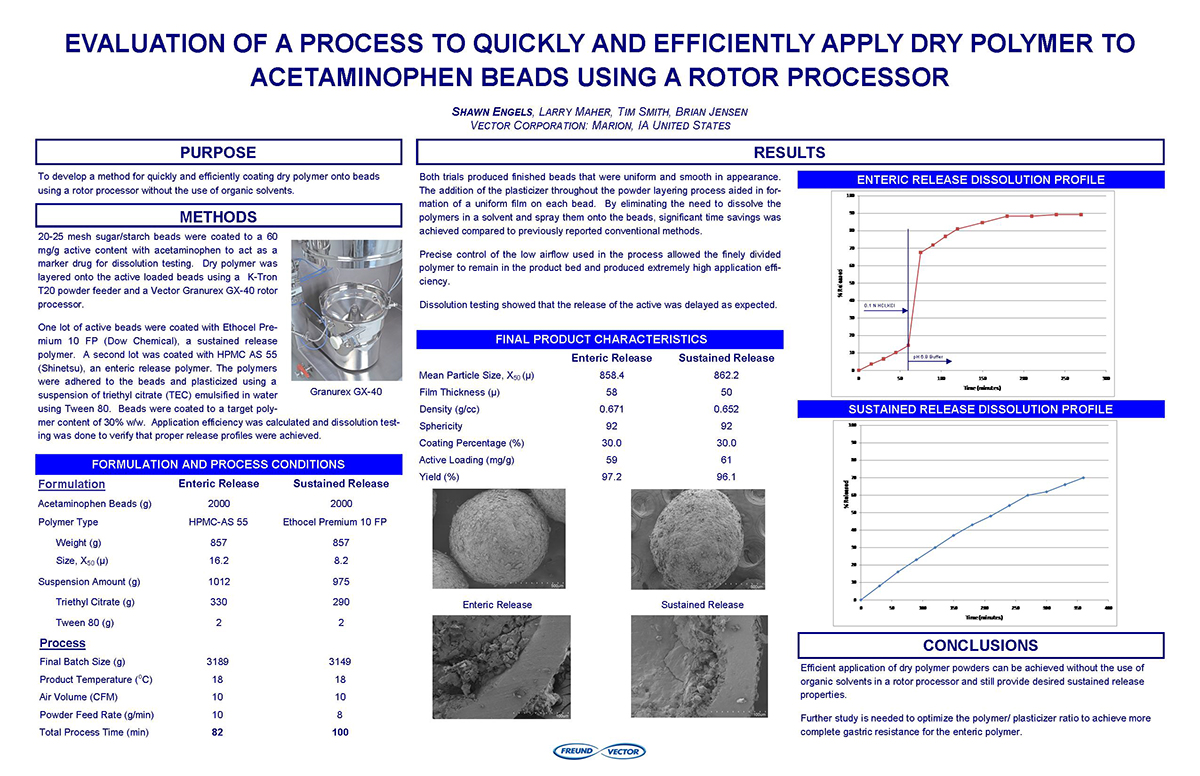
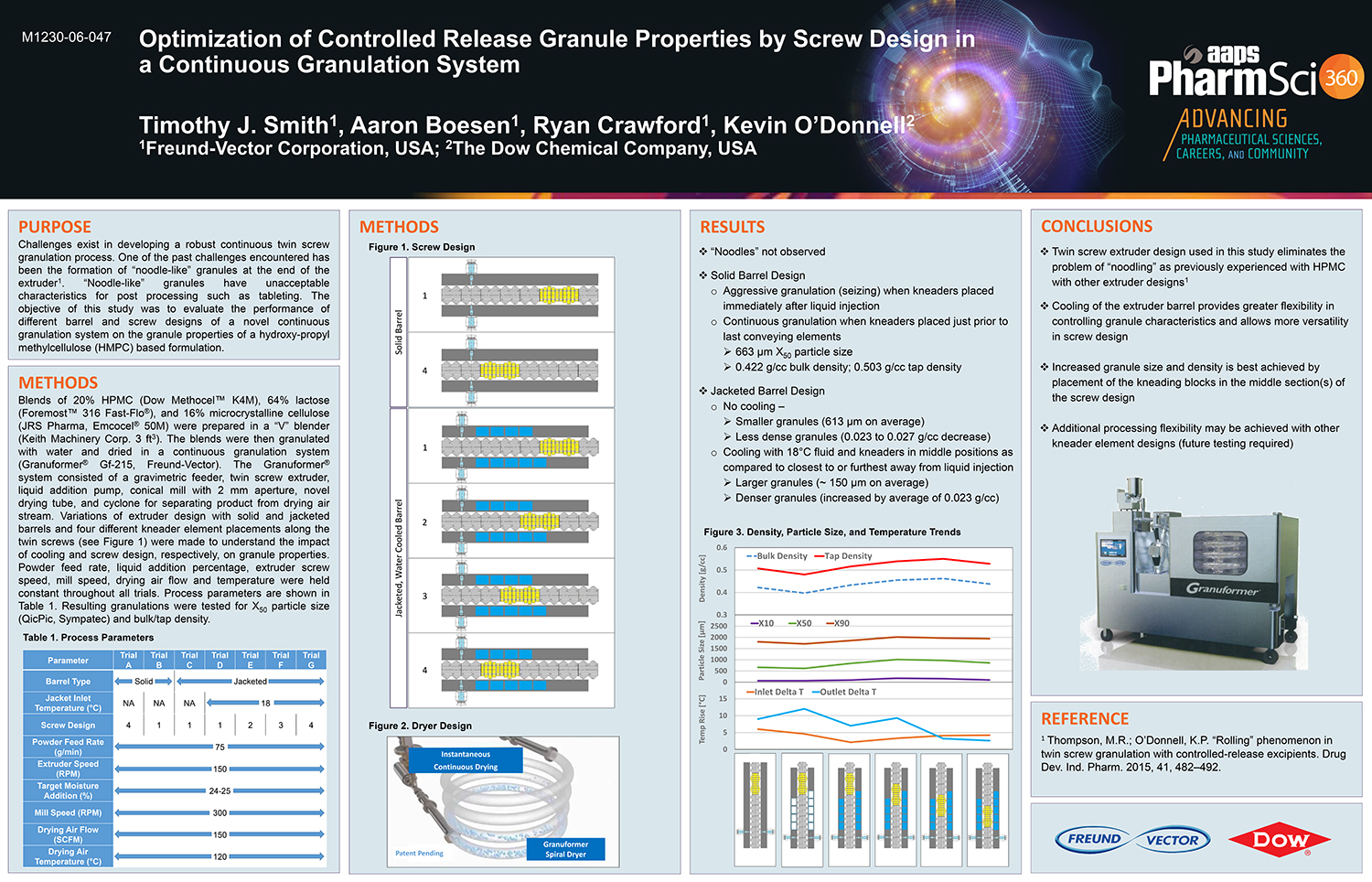
Bottom Spray Wurster technology is commonly used in the pharmaceutical industry as a method for applying active and functional coatings, including sustained release and enteric polymer coatings, to multi-particulate substrates. Typically aqueous dispersion of polymers are diluted and are mixed with appropriate glidant, such as talc, to reduce blocking and agglomeration during the drying of the polymer solution on the surface of the multi-particulate. These solutions and suspensions are applied via air atomizing spray guns. The need to dilute the solution can increase the application time needed for proper drug release or enteric protection.